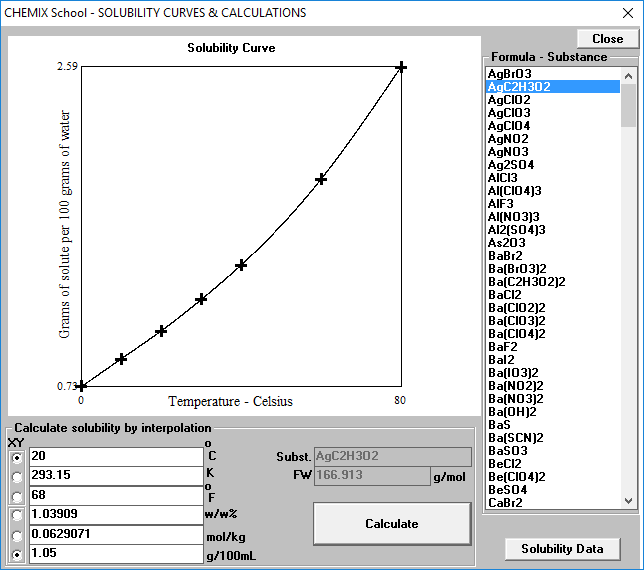
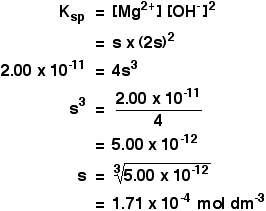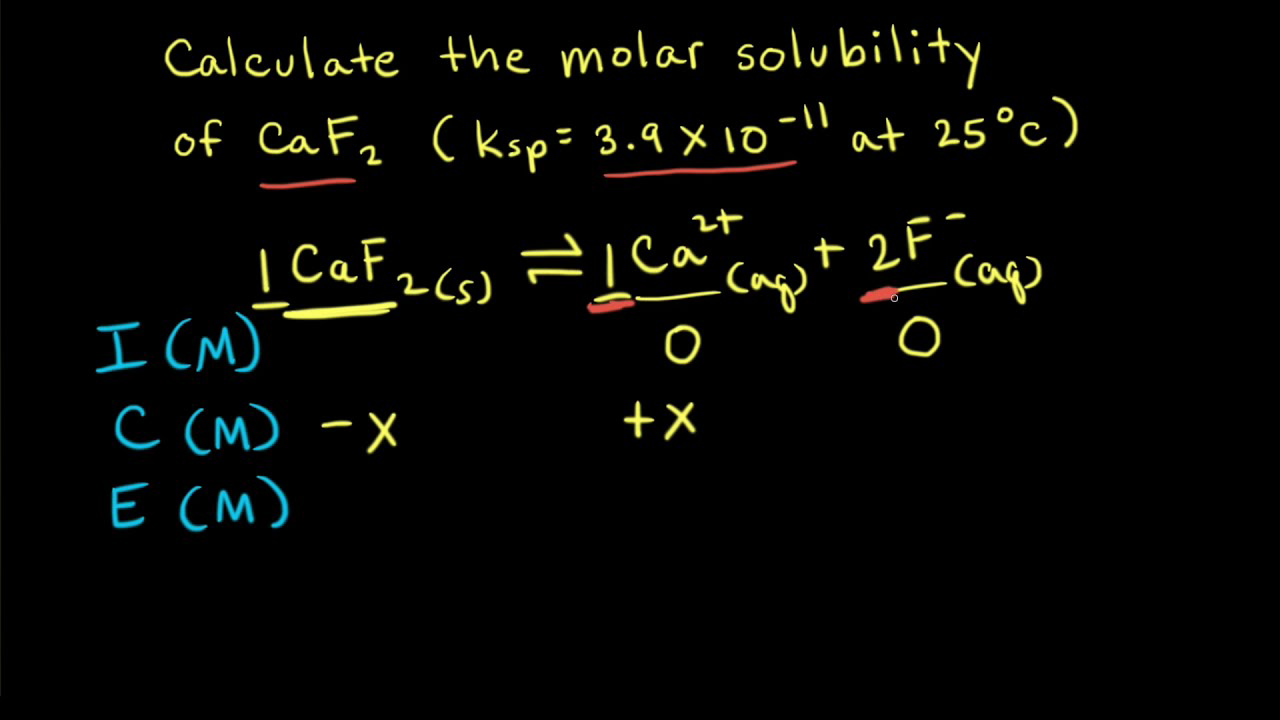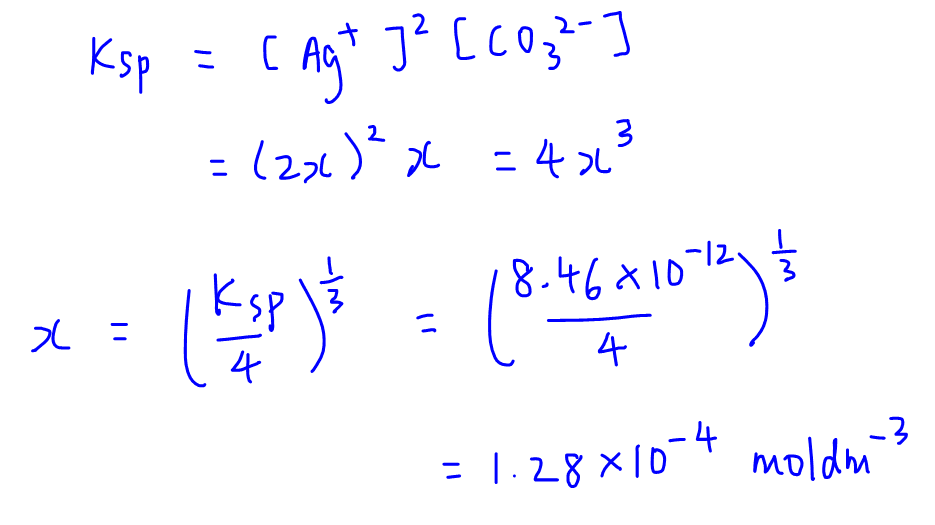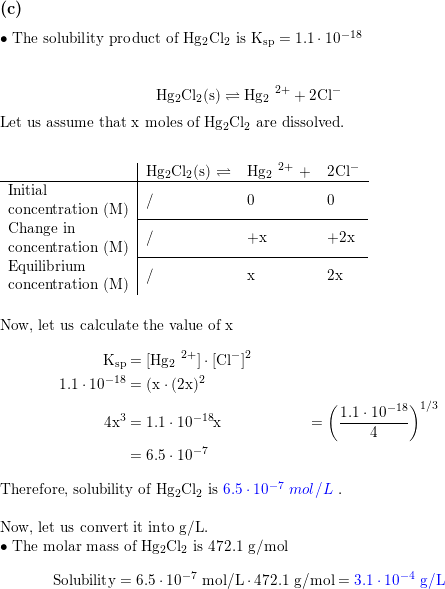✓ Solved: Calculate the molar solubility of SrC2O4 in a solution that has a fixed H3O^+ concentration...

SOLVED: Calculate the solubility (in g/L) of O2 in water at a partial pressure of O2 of 120 torr at 25C (K o2 = 780 atm*L/mol)
How to Calculate and Solve for Glass Gas Solubility Parameter in a Fluid | Nickzom Calculator - Nickzom Blog